


Complementary to crossed molecular beam experiments at the University of Hawai’i, the elementary reaction mechanisms leading to the formation of polycyclic aromatic hydrocarbons (PAHs) are explored within a high temperature chemical reactor by probing isomer distributions via tunable vacuum ultraviolet (VUV) radiation from the Advanced Light Source (ALS), Swiss Light Source (SLS), and SOLEIL Synchrotron in collaboration with Dr. Musahid Ahmed (ALS), Dr. Patrick Hemberger (SLS), and Dr. Laurent Nahon (SOLEIL). So far, molecular beam experiments in conjunction with electronic structure calculations by Prof. Mebel (Florida International University) unraveled via five elementary mechanisms leading to PAHs: Hydrogen Abstraction – Acetylene Addition (HACA), Hydrogen Abstraction – Vinylacetylene Addition (HAVA), Phenyl Addition – DehydroCyclization (PAC), Radical-Radical Reactions (RRR), and Methylidyne Addition - Cyclization - Aromatization (MACA). These concepts provide a solid, experimentally and computationally constrained framework to PAHs in extreme environments from low temperatures in molecular clouds (10 K) and hydrocarbon-rich atmospheres of planets and their moons (35–150 K) to high temperature environments like circumstellar envelopes of carbon rich AGB stars and combustion systems at a few 1,000 K. The chemical microreactor presents a versatile approach to study the formation of combustion-relevant polycyclic aromatic hydrocarbons (PAHs) under well-defined and controlled conditions eventually leading to 2D and 3D nanostructures such as nanosheets and fullerenes in extreme environments. Experiments are currently in preparation to explore the mainly elusive outcome of radical–radical reactions leading to PAHs carrying between two and four rings and to molecular wires along with nanobelts carrying multiple benzene rings. Further, we aim to increase the level of molecular complexity by untangling the pathways to carbonaceous buckybowls and buckyballs.
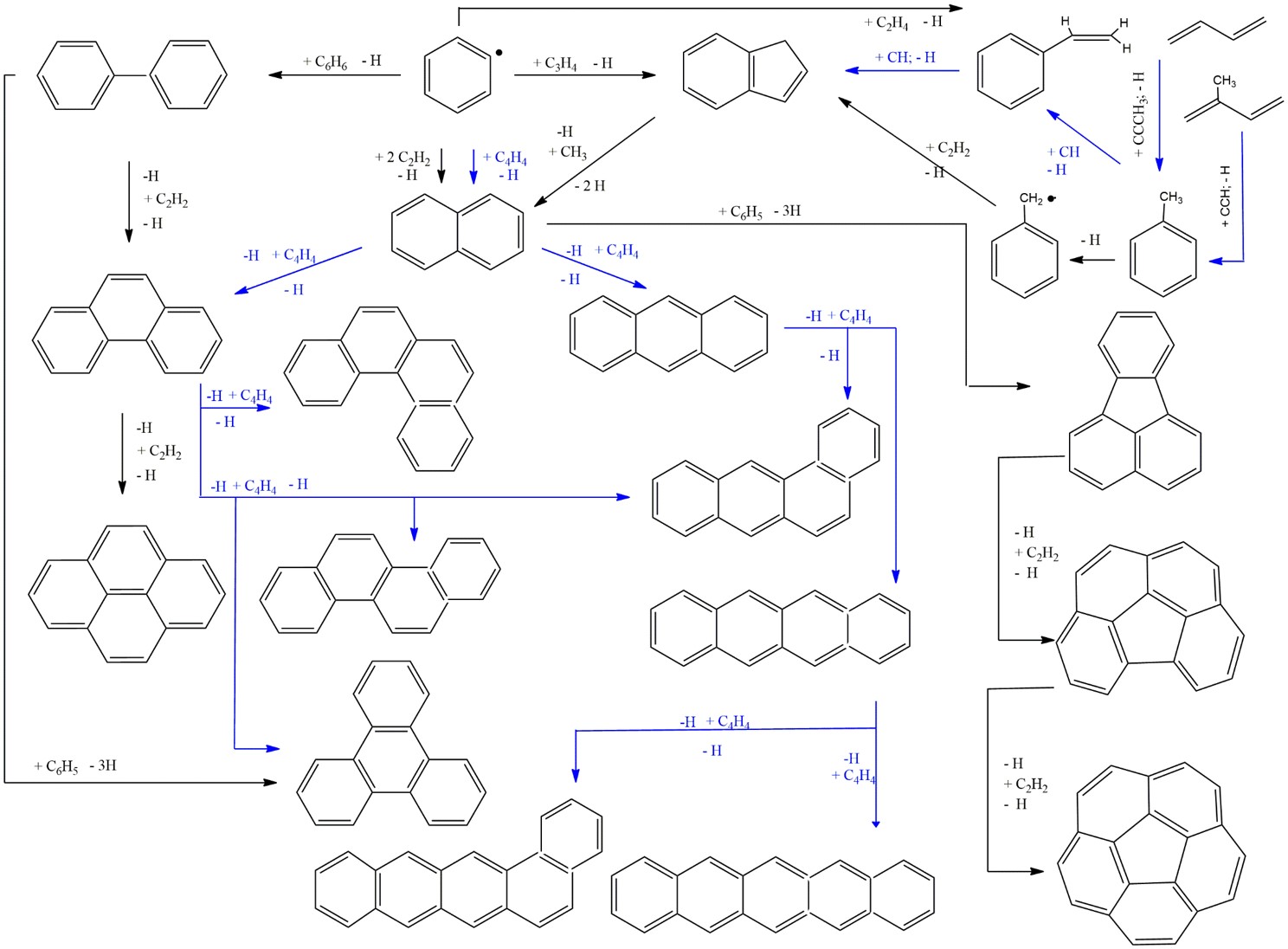
Selective molecular mass growth processes leading to aromatic systems with up to six rings based on our studies. Pathways color coded in blue represent barrierless reaction mechanisms. Multiple pathways leading to identical products like naphthalene, triphenylene, and indene highlight the complexity of prospective chemical reaction networks for combustion systems and in extraterrestrial environments.